TIP4P model of water: Difference between revisions
Carl McBride (talk | contribs) m (→Parameters) |
Carl McBride (talk | contribs) m (Added mention of second virial coefficient) |
||
| (One intermediate revision by the same user not shown) | |||
| Line 15: | Line 15: | ||
==Phase diagram== | ==Phase diagram== | ||
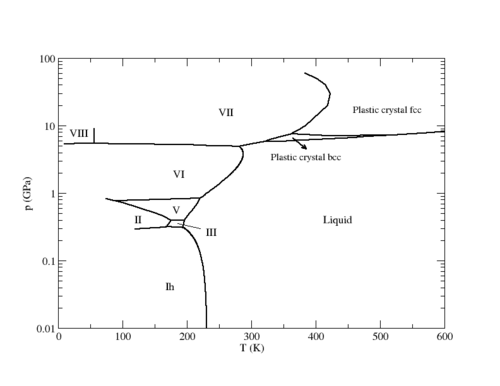
The following is the phase diagram of the TIP4P model in the [[Phase diagrams: Pressure-temperature plane |pressure-temperature plane]] (adapted from Fig. | The following is the phase diagram of the TIP4P model in the [[Phase diagrams: Pressure-temperature plane |pressure-temperature plane]] (adapted from Fig. 11 of <ref name="multiple2">[http://dx.doi.org/10.1063/1.3156856 J. L. Aragones and C. Vega "Plastic crystal phases of simple water models", Journal of Chemical Physics '''130''' 244504 (2009)]</ref>). | ||
:[[Image:TIP4P_phase_diagram.png|500px]] | :[[Image:TIP4P_phase_diagram.png|500px]] | ||
===Plastic crystal phases=== | ===Plastic crystal phases=== | ||
| Line 24: | Line 24: | ||
==Shear viscosity== | ==Shear viscosity== | ||
The [[shear viscosity]] for the TIP4P model is 0.494 mPa.s at 298 K and 1 bar <ref>[http://dx.doi.org/10.1063/1.3330544 Miguel Angel González and José L. F. Abascal "The shear viscosity of rigid water models", Journal of Chemical Physics '''132''' 096101 (2010)]</ref> (experimental value 0.896 mPa.s <ref>[http://dx.doi.org/10.1021/je049918m Kenneth R. Harris and Lawrence A. Woolf "Temperature and Volume Dependence of the Viscosity of Water and Heavy Water at Low Temperatures", Journal of Chemical & Engineering Data '''49''' pp. 1064-1069 (2004)]</ref>). | The [[shear viscosity]] for the TIP4P model is 0.494 mPa.s at 298 K and 1 bar <ref>[http://dx.doi.org/10.1063/1.3330544 Miguel Angel González and José L. F. Abascal "The shear viscosity of rigid water models", Journal of Chemical Physics '''132''' 096101 (2010)]</ref> (experimental value 0.896 mPa.s <ref>[http://dx.doi.org/10.1021/je049918m Kenneth R. Harris and Lawrence A. Woolf "Temperature and Volume Dependence of the Viscosity of Water and Heavy Water at Low Temperatures", Journal of Chemical & Engineering Data '''49''' pp. 1064-1069 (2004)]</ref>). | ||
==Virial coefficients== | |||
The [[second virial coefficient]] has been calculated by Chialvo et al <ref>[http://dx.doi.org/10.1016/j.molliq.2006.08.018 Ariel A. Chialvo, Albert Bartók and András Baranyai "On the re-engineered TIP4P water models for the prediction of vapor–liquid equilibrium", Journal of Molecular Liquids '''129''' pp. 120-124 (2006)]</ref>. | |||
==See also== | ==See also== | ||
The following is a list of [[water models | empirical models]] that build upon this TIP4P model: | The following is a list of [[water models | empirical models]] that build upon this TIP4P model: | ||
Latest revision as of 14:15, 20 January 2011
The TIP4P model [1] is a rigid planar four-site interaction potential for water, having a similar geometry to the Bernal and Fowler model, which was proposed in 1933.
Parameters[edit]
The TIP4P model consists of a Lennard-Jones site for the oxygen atom, and three charge sites.
| (Å) | HOH , deg | (Å) | (K) | q(O) (e) | q(H) (e) | q(M) (e) | (Å) |
| 0.9572 | 104.52 | 3.154 | 78.0 | 0 | 0.52 | -2q(H) | 0.15 |
Phase diagram[edit]
The following is the phase diagram of the TIP4P model in the pressure-temperature plane (adapted from Fig. 11 of [2]).
Plastic crystal phases[edit]
Recent simulations have demonstrated the existence of plastic crystal phases for the TIP4P model [2].
Melting point[edit]
The melting point ice Ih-water at 1 bar is [3].
Shear viscosity[edit]
The shear viscosity for the TIP4P model is 0.494 mPa.s at 298 K and 1 bar [4] (experimental value 0.896 mPa.s [5]).
Virial coefficients[edit]
The second virial coefficient has been calculated by Chialvo et al [6].
See also[edit]
The following is a list of empirical models that build upon this TIP4P model:
- TIP4F
- TIP4P
- TIP4P/2005
- TIP4P-Ew
- TIP4P/FQ
- TIP4P-HB
- TIP4P/Ice
- TIP4P-pol
- TIP4PQ/2005
- TIP4P-QDP
- TIP4P-QDP-LJ
References[edit]
- ↑ William L. Jorgensen, Jayaraman Chandrasekhar, Jeffry D. Madura, Roger W. Impey and Michael L. Klein "Comparison of simple potential functions for simulating liquid water", Journal of Chemical Physics 79 pp. 926-935 (1983)
- ↑ 2.0 2.1 J. L. Aragones and C. Vega "Plastic crystal phases of simple water models", Journal of Chemical Physics 130 244504 (2009)
- ↑ Carlos Vega, Maria Martin-Conde and Andrzej Patrykiejew "Absence of superheating for ice Ih with a free surface: a new method of determining the melting point of different water models", Molecular Physics 104 pp. 3583-3592 (2006)
- ↑ Miguel Angel González and José L. F. Abascal "The shear viscosity of rigid water models", Journal of Chemical Physics 132 096101 (2010)
- ↑ Kenneth R. Harris and Lawrence A. Woolf "Temperature and Volume Dependence of the Viscosity of Water and Heavy Water at Low Temperatures", Journal of Chemical & Engineering Data 49 pp. 1064-1069 (2004)
- ↑ Ariel A. Chialvo, Albert Bartók and András Baranyai "On the re-engineered TIP4P water models for the prediction of vapor–liquid equilibrium", Journal of Molecular Liquids 129 pp. 120-124 (2006)